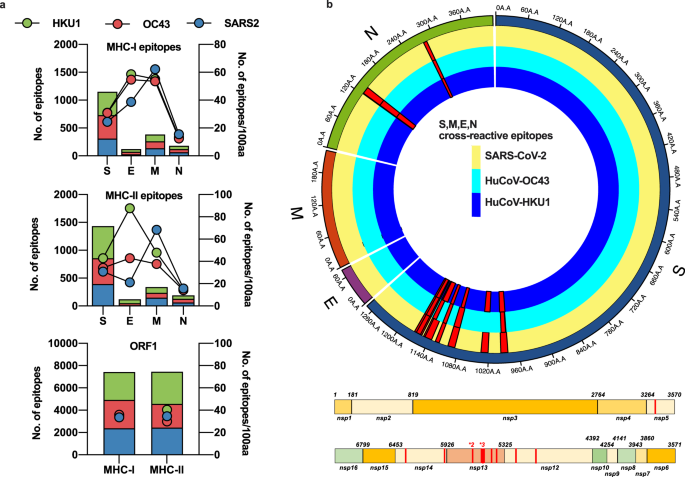
drron
Veteran Member
- Joined
- Jul 4, 2002
- Posts
- 37,774
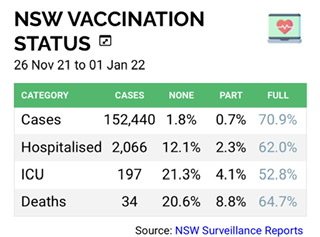
There are also figures from Israel that are unkind to Pfizer.despite rolling out the fourth dose of Pfizer in the last week they have just had the highest daily cases numbers of the pandemic -75000 cases. Equal, when population differences are taken into account, of 210000 cases in Australia per day.John Cambell of Youtube fame - well there are provisional UK figures that are unkind to AZ and Omicron. Is the acid test serious/ICU, moderate, or just transmissible? I believe serious or death is well covered if vaccinated. Both WA and China are on elimination strategy. And US is saying don't go to Spain which has higher vaccination rates than Au. The funny thing about USA is they had their shots early, so there just might be a U or double Bell curve for resistance levels. I wonder if long term vaccine hamsters/ GM ACE2 mice are being monitored for effects over time? My gut feeling is those >80-85 and or with co-mortality are the priority , adjusted for new treatment/drugs that we did not have before. It remains to be seen if nursing homes and the like have lifted their game.
We just have to face the fact that with Omicron none of the existing vaccines are going to be the way out. Covid zero is now a failed policy if it wasn't already.