lovetravellingoz
Enthusiast
- Joined
- Jul 13, 2006
- Posts
- 12,745
covidlive is now estimating that VIC is just a week away from 80% first dose, a day after the VIC premier estimates it will be reached. If vaccination rates pick up a bit this week the Premier's estimate could prove correct.
Hopefully with the Moderna the Feds will be disprortianatly directed it to the Vic Hotspot LGAs as requested. Ditto with Pfizer to the GPs. Recently the Commonwealth Channel was still mainly vaccinating in non-hotspot LGA's.
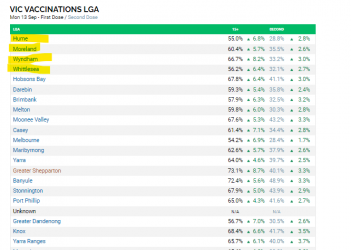
While the Vic State Average is 72.8% first dose and 43.9% second dose, it is higher in the country regions and lower in Metro Melbourne, and significantly lower in the suburbs with the highest daily case numbers.
So while 80% statewide is not far off, getting the suburbs with the highest cases is going to take longer and in terms of controlling the case rate and hospitalisation rate this is most important.
Cases still dominate in 4 LGA's and mainly in the City of Hume. (See below) In these LGA's and also in the other LGA's with significant case numbers the vaccination rate tends to be much lower than the state average.
City of Hume with the largest number of daily cases has a first dose rate of only 55%. This went up 6.8% last week, but this is still way to low for such an epicentre of cases. So it is vital that City of Hume in particular gets that 6.8% weekly increase rate increased.
The State Hubs and State pop-ups (some are located at schools, others places of worship) are more focussed now on residents of the LGA's with higher case rates including City of Casey in the SE.
But the Commonwealth run GP's and Pharmacists are disprortianatly not in the hotspot suburbs. More are meant to opening this week, and over the next four. So hopefully doses will be going where they are needed most can rapidly change. But it will probably be October before a decent number of GP's and Pharmacists are actually operating in Vic.
Breakdown of Victoria's 567 new cases and vaccination rates
COVID commander Jeroen Weimar says the age profile of the cases are largely younger people, which is a familiar pattern.
"[Of the new cases] 82 per cent are aged under 50."87 per cent of all cases are in the northern and western suburbs of Melbourne. In the north, 357 cases — including Craigieburn, Roxburgh Park and Broadmeadows — and a quarter of the cases are in the western suburbs."There are also 13 cases in regional Victoria and two further cases following up and we will can firm those this afternoon."Over half of our cases today are in households where they have been previous cases."
Last edited: